
Buffalo flies can cause production losses due to lower weight gain, lower milk yield and permanent hide damage.
BUFFALO flies are ranked as the number one economic pest of cattle in Australia, costing the cattle industry $111.7 million per annum in production losses, and a further $60m annually in control measures.
The blood-feeding insects cause significant irritation to cattle and result in skin lesions, as well as decreased milk and meat production.
And the problem is only getting worse, with modelling predicting the range of persistent populations will expand further towards the southern states in coming years.
The recent loss of Diazinon as an approved active ingredient for the control of buffalo fly in Australia – due to critical safety risks, environmental concerns and potential trade non-compliance as ruled by the Australian Pesticides and Veterinary Medicines Authority (APVMA) in 2025 – has further reduced the tools the industry can draw upon to effectively control the biting insects.
A recent Queensland DPI survey has further highlighted the extent of the problem, with 87 percent of 177 participating producers confirming they relied on ear tags, pour-ons and sprays to treat herds for buffalo fly, and 56pc observing declines in the effectiveness of insecticides at controlling buffalo flies.
Resistance is a genetic trait that allows insects to survive insecticide treatments and is heritable from parent to offspring.
But as a number of research papers presented at the Northern Australian Beef Research Update Conference (NBRUC) in Brisbane this week demonstrated, researchers are not tackling the problem lying down.
Promising developments showcased at NBRUC included the identification of specific genetic mutations responsible for insecticide resistance in buffalo flies.
The conference also heard encouraging in-paddock results from tests of a soon-to-be-launched new insecticidal ear tag in Australia, which will introduce a new active ingredient to northern producers’ armoury (more below)
Genetic mutation behind buffalo fly resistance pinpointed
Dr Jess Morgan from Queensland DPI outlined recent research to identify the cause of buffalo resistance to insecticide.
To identify if levels of resistance were increasing, the researchers used the “classic method” of exposing buffalo flies to filter papers impregnated with different dosages of insecticide.
If flies die after exposure they are susceptible; if they survive they are resistant.
What researchers found came as ‘a bit of a shock’
They found that flies were surviving at “hundreds of times” the discriminating dose.
Researchers also noticed some flies that appeared on the brink of death after initial exposure were up and walking again 24 hours later.
This represented the first time “knockdown recovery” had been observed in buffalo flies.
This came as a “bit of a shock”, Dr Morgan told the audience.
“We don’t run the assays that long (usually two to four hours, not 24 hours). So that was a trigger for this research. What was going on?”
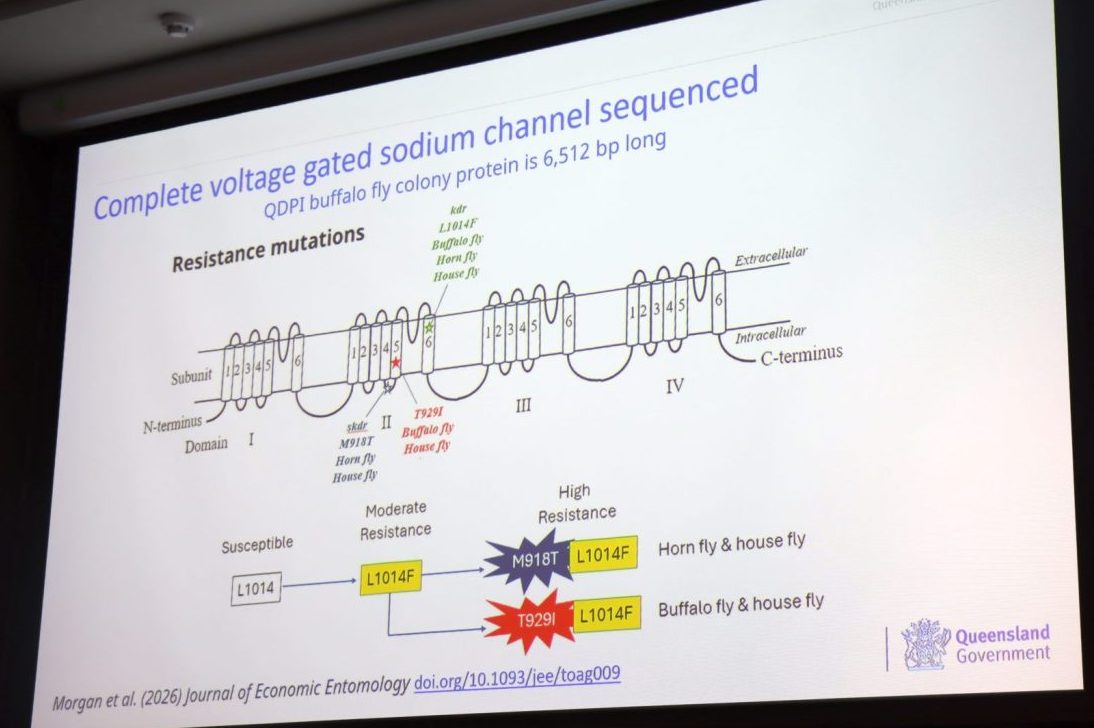
Suspecting that mutations in the pores of flies – which consist of a ‘voltage-gated sodium channel’ (VGSC) – may be conferring resistance, DNA was extracted from the DPI buffalo fly colony and screened for resistance-linked mutations.
Two mutations were identified that that the scientists believe explain 82 percent of cypermethrin and deltamethrin resistance in flies. Flies with both mutations had levels of “super resistance”.
“It’s the reason why you’re having to put a lot more chemical on to get an effect,” Dr Morgan said.
“This work, I’m very pleased to say, just got published about a month ago. It’s now out there. If you’re interested in more, there’s plenty more in that paper.”
Dr Morgan said the research team believes the “knockdown recovery” now being witnessed may be linked to detoxifying enzymes, which follow a different pathway to the voltage-gated sodium channel.
“What’s worrying about it is that your insecticide appears to be working – the flies are getting knocked off the animals – but as soon as they’re no longer exposed to chemical, they’re coming back to life and flying off.
“So they’re recovering, so that is a concern.”
On the plus side, evidence from other insect groups suggested that adding a synergist to an insecticide may help block detoxifying pathways.
“We really want to investigate that a bit more to figure out whether we can improve improve the situation of some of these chemicals.”
Future research will investigate whether adding a synergist could boost the performance of chemicals that appear to be failing, as well as further work on non-chemical control options such as buffalo fly tunnel traps.
“This is research that was done back in the 2000s. DPI released a build your own tunnel trap,” Dr Morgan said.
“All of the research, all the science behind those tunnel traps say they work, but uptake has been quite low.
“The survey said about 7 percent of people are using them, but all the feedback is still that they work.
“So we think there’s been a bit of a falling down in the promotion of it and the extension.”
Promising results for a new type of insecticidal ear tag
Tim Schatz, Director of Livestock Industries at NT DAF, also presented buffalo fly related research highlighting positive results from a recent trial testing a new ear tag developed by Y-Tex Corporation.
 The tag contains a new combination of active ingredients, one of which represents a new class of active ingredient for the buffalo fly tag market in Australia.
The tag contains a new combination of active ingredients, one of which represents a new class of active ingredient for the buffalo fly tag market in Australia.
Two groups of Brahman heifers at the Beatrice Hill Research Station in the Northern Territory – a floodplain site with “lots of water and lots of buffalo flies” – were randomly allocated to either a control group, which received no treatment for fly control, or a treatment group, which was fitted with the new sustained-release plastic tags.
The heifers had previously been grazed as one mob, but during the trial period were run in separate but similar neighbouring paddocks with access to mineral lick blocks and managed the same way.
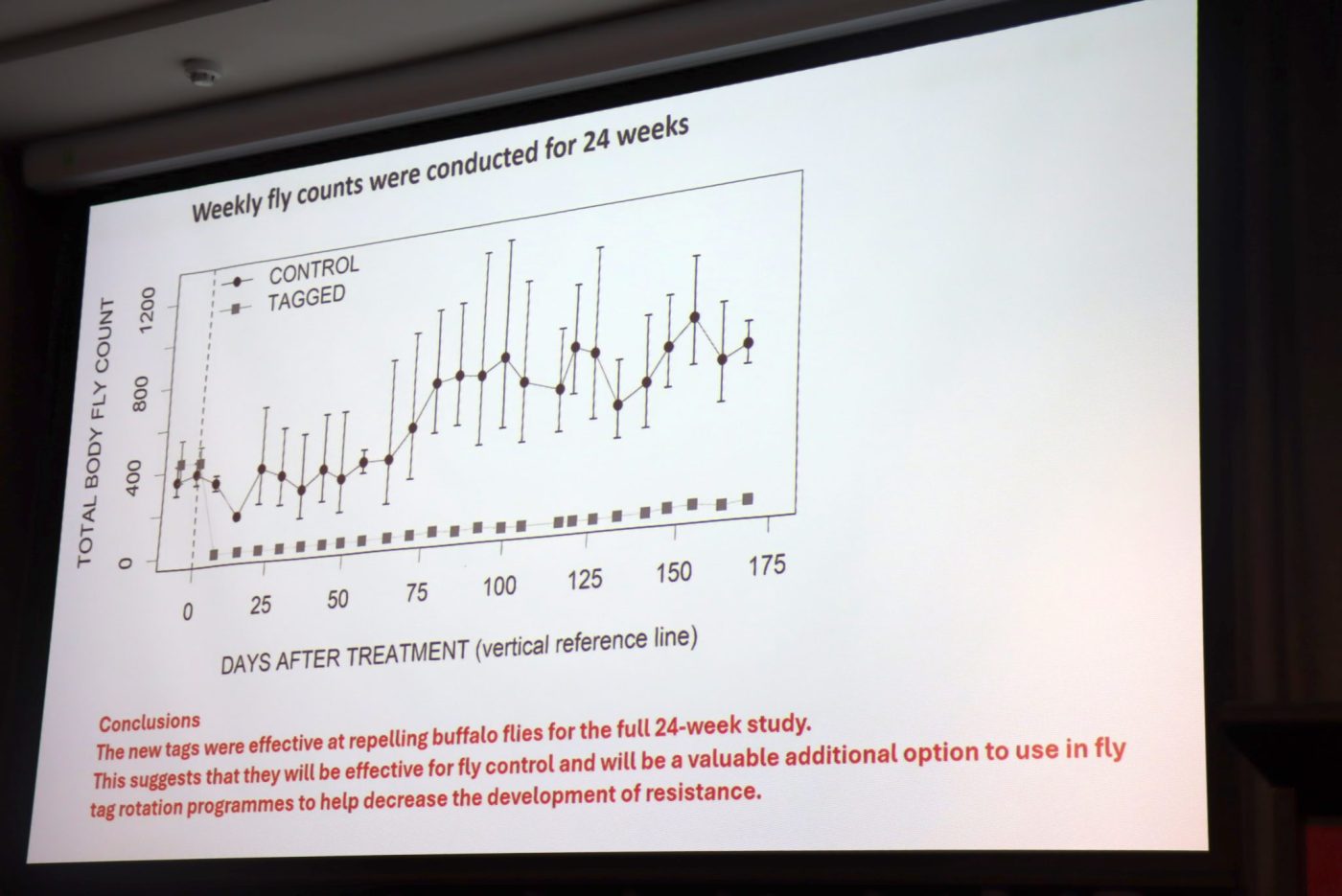
Fly counts were conducted seven days prior to the start of the trial and then weekly for 24 weeks.
The number of flies on the control group ranged from about 500 to 700 throughout the trial, peaking at about 800 per animal in week 22.
In contrast, fly counts on the tagged treatment group remained significantly lower, staying at less than 10–15 flies per animal until week 21 and remaining below 25 until the trial finished after 24 weeks.
“So these results suggest that this new tag is very effective in repelling flies, and will be quite useful for use in rotations to prevent the development of resistance in buffalo flies to fly tags,” Mr Schatz said.


HAVE YOUR SAY